-
甲基叔丁基醚(methyl tert-butyl ether, MTBE)是一种广泛使用的汽油添加剂,分子中的叔碳原子和甲基结构,使MTBE有良好的化学稳定性。随着国内汽油消费量的快速增长,MTBE产量持续增加,在世界范围内达到了约每年20 Mt [1]。MTBE在欧美等国已经被禁止使用,但由于储油罐、输送管道及加油站汽油的意外泄露造成环境中的残留污染依然存在。因具有相对较高的溶解度(42 g·L−1, 20℃)和较低的亨利系数(5.87×10−4 atm·m−3·mol−1, 25℃)[2],在环境中易溶于水且不易被土壤颗粒吸附[3]。由于MTBE的挥发性和难降解性,其自然衰减速率较低,能够在环境中持久存在并造成水体污染[4]。MTBE是浅层地下水中排名第二的常见挥发性有机化合物[5],可明显改变水体的气味和味道[6]。美国环境保护署(Environmental Protection Agency, EPA)建议饮用水中MTBE的味觉和气味阈值分别为40 、20 µg·L−1[7]。研究表明,MTBE具有遗传毒性并对皮肤和眼睛具有刺激性,在高浓度下还有抑制神经系统的可能性[8]。此外,MTBE会引起多种类型的DNA损伤,例如单链、双链断裂等[9]。由于MTBE污染的普遍性及潜在毒性,EPA在2000年将其划分为危害健康的物质[10],又在2002年通过了于2006年实施禁用MTBE的议案[11]。随着对MTBE毒性认识的深入,我国提出了在2020年禁用MTBE的环境规划[12]。尽管囿于技术发展水平实施该规划尚有较大难度,但治理环境中潜在的MTBE污染已是目前一大重要课题。
MTBE的分子结构:

基于MTBE对环境产生的污染及对人体健康造成的危害,研究人员对其降解技术进行了大量研究。常见的处理方法包括空气汽提、物理吸附、高级氧化及生物降解[5]。Vignola等[13-14]利用原位可渗透性反应墙修复被MTBE和烃类污染的地下水,经过约100 d的处理将MTBE的浓度降到了10 µg·L−1以下。Burbano等[15]用芬顿法处理MTBE,反应1 h后降解率达到了约99%。Salanitro等[16]培养的混合菌种BC-1是最早报道的能够降解MTBE的微生物。自此,通过研究发现了更多能以MTBE作为唯一碳源和能源进行利用,或通过共代谢方式将其降解的混合菌群和单一菌株[17-18]。
尽管已经证实上述处理方法均能在一定程度上降解MTBE,但每种单独的修复技术都存在固有的缺点。其中,空气汽提法在去除较低浓度的MTBE时会消耗大量的空气流,需要搭配尾气处理装置以净化被污染的空气流,技术成本较高[5]。吸附技术会受到吸附容量的限制,且反应后MTBE仍存在于吸附剂中会产生二次污染,存在废吸附剂的处理问题。此外,地下水中的天然有机物会产生竞争吸附作用而降低处理效果[19]。高级氧化法的成本较高,会产生毒性更大的副产物,易造成二次污染[20]。对于生物处理,MTBE分子结构中的醚键及叔丁基使其具有较低的生物降解性[21]。且该方法需要投放微生物会影响当地的微生物生态,并会受到环境中温度、pH、营养物质浓度等因素的限制,在原位修复中仍有较多问题亟待解决[22]。
为了克服上述修复技术单独使用时各自的局限性,研究人员开始探索将不同的技术联合应用,通过协同效应以提高降解效率。目前,已发表有大量的相关研究详细介绍不同类型的联合修复技术。基于吸附技术具有工艺控制和操作简单;高级氧化技术处理效率高;生物降解法不会产生二次污染的特点,本文选择以上3种处理技术,总结并概述了在这3种处理方法之中应用不同的联合修复技术去除水中MTBE污染的反应机理及应用,表1归纳了各技术的优缺点。
-
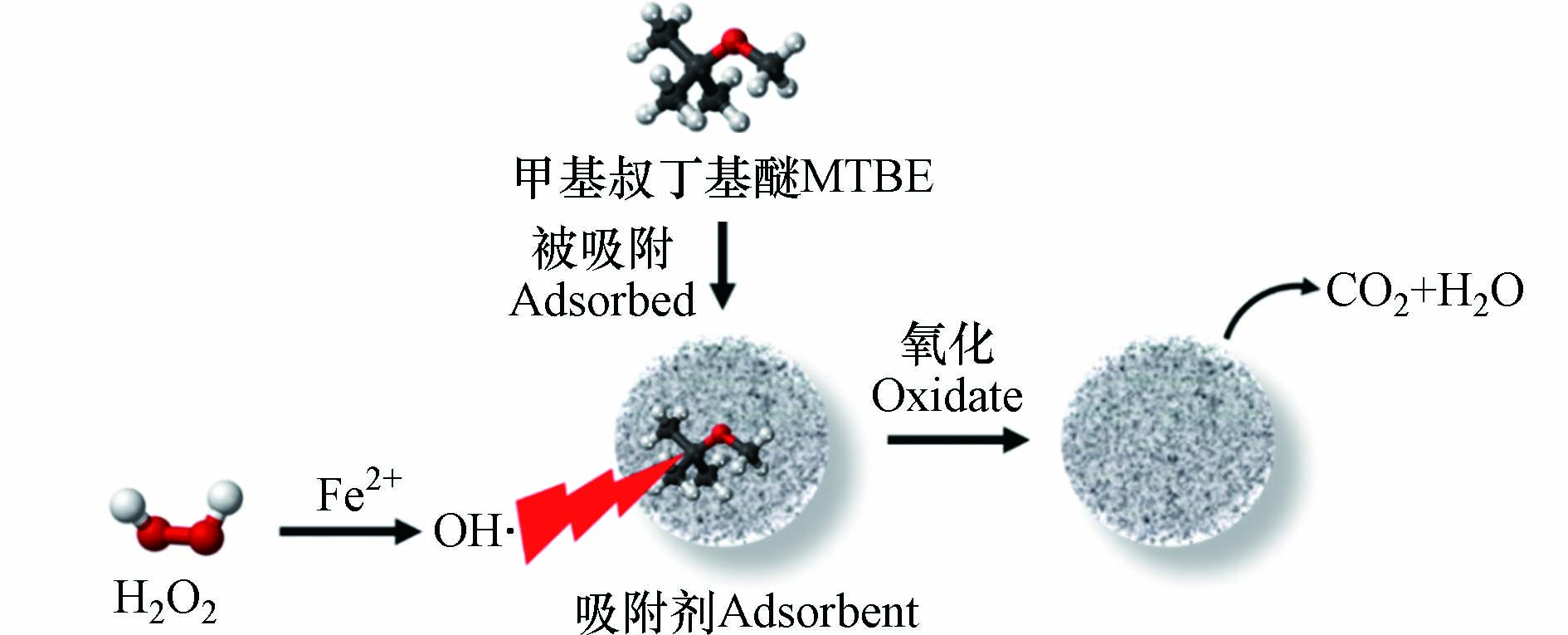
该方法借助吸附剂将MTBE吸附并富集到反应位点附近,再利用产生的活性自由基完成氧化降解反应[23-24],其机理示意图如图1所示。活性自由基可以通过多种方式产生,例如,通过Fenton反应生成的羟基自由基(OH·)是仅次于氟的强氧化剂,氧化能力极强。OH·与MTBE之间的反应速率常数很高(1.6×109 L·mol−1·s−1),表明MTBE容易受到OH·的攻击被氧化降解,其反应机制总结如下[25-26]:
还可以通过紫外辐射以光化学的方式形成[27-28],运用电化学技术在电极通过电化学反应形成[29- 30],或利用光芬顿法,其反应机制总结如下[31-32]:
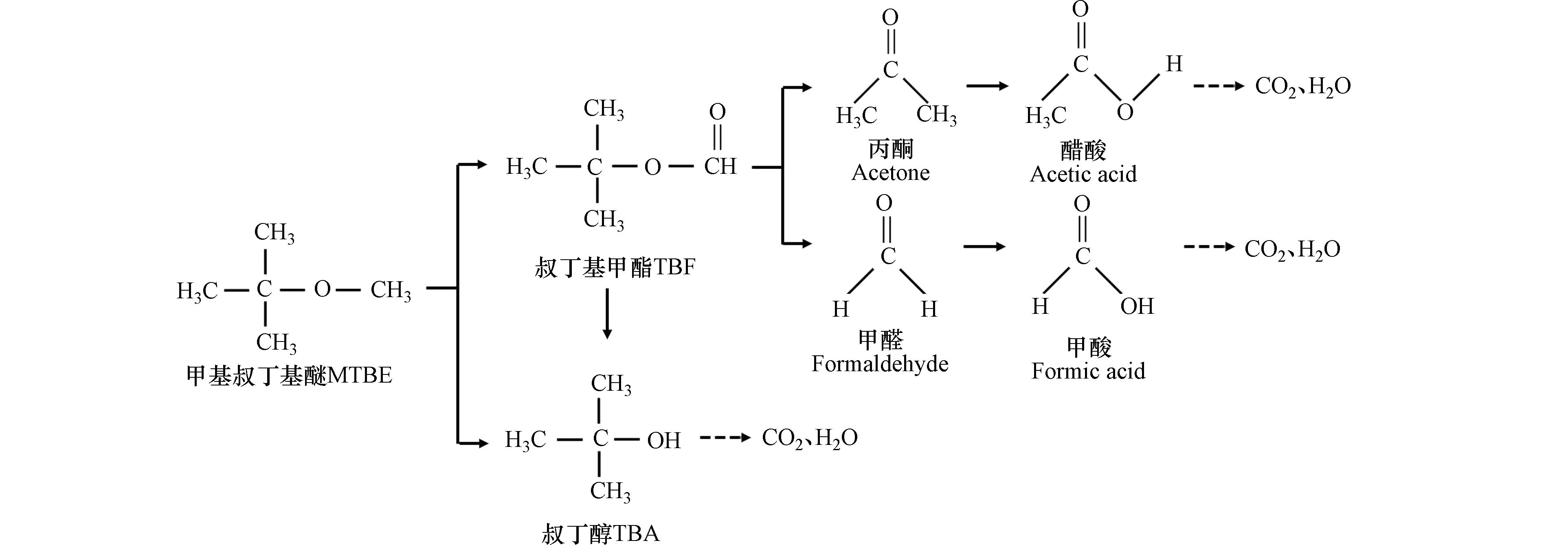
Sedlazeck等[33]的实验证实,将上述几种技术联合应用可形成更多的OH·以显著提高反应速率。近年来,研究人员对OH·氧化降解MTBE的反应途径进行了深入探索[15, 34],可能的降解途径如图2所示。图2中实线表示可观测的变化,虚线表示可得到产物但代谢机理尚不清楚。考虑到OH·的电荷分布情况和亲电特性,在反应过程中首先攻击MTBE结构中的甲氧基,而后转化为叔丁醇(tert-butyl alcohol, TBA)和叔丁基甲酯(tert-butyl formate, TBF)。实验证明,TBA及TBF是可被直接检测到的主要的中间产物,能被进一步降解[15]。其中,TBF结构中的甲酰基更容易受到OH·的攻击而生成HCOOH和CH3COOH,最终经过一系列反应被完全氧化降解为CO2和H2O。
-
对于MTBE常用的吸附材料有沸石、活性炭等。沸石具有多种特性:(i)作为硅酸铝材料,有较好的抗氧化性能避免被氧化降解,且可通过多种方式实现对沸石的再生处理[35-36],使用寿命较长;(ii)离子交换能力较强而易实现铁的掺杂;(iii)对目标污染物的吸附亲和力及吸附容量取决于被吸附物质和沸石的疏水性以及分子和孔径大小的一致性;(iv)具有较高SiO2/Al2O3的含铁沸石对于吸附小分子有机物(如MTBE)有天然的优势[23, 37]。研究证明,该联合技术对目标污染物的吸附富集效应对降解是有益的[38],常用的沸石材料主要包括ZSM-5、Beta、八面沸石、丝光沸石等。
ZSM-5是一种硅含量高,孔隙大小略小于容纳MTBE的最适分子大小,疏水性好的MFI型沸石[23]。环境pH值对其吸附能力没有明显影响,是对MTBE吸附效率最高的材料之一,可以用于复杂的自然水环境中[39]。例如,腐殖酸是地下水中普遍存在的一种天然有机物(Natural Organic Matter, NOM)。实验表明,在较高浓度腐殖酸(100 mg·L−1)存在的条件下对MTBE的降解率并没有明显影响[38]。这与相关研究所论证的ZSM-5在抵抗NOM对吸附性能的干扰方面有明显优势的结论是一致的[37, 40]。其原因在于腐殖酸分子较大而难以进入沸石孔隙结构,在很大程度上降低了其与OH·发生反应的可能性,即ZSM-5可适用于含有中高浓度腐殖酸的水环境。
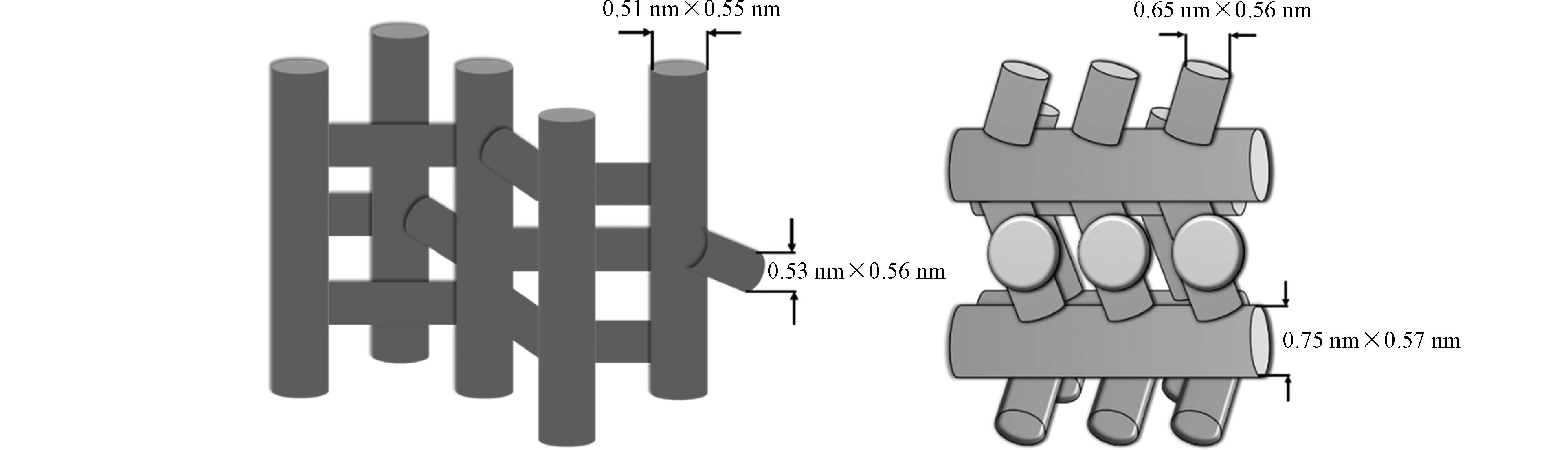
对ZSM-5与Beta两种类型沸石的性能进行比较。一方面,Beta的吸附亲和力高于ZSM-5,其原因如下。首先,SiO2/Al2O3较高的沸石在其孔隙内具有疏水性表面,水无法在疏水性高的沸石的孔径结构中形成冷凝液相[41-42]。如表2所示,选择物理性质相似的两类沸石,根据SiO2/Al2O3推测Beta的疏水性更高,因此其对MTBE的吸附亲和力更强。其次,这两种沸石的孔径明显不同[24]。如图3所示,ZSM-5具有10环通道结构(正弦通道和直通道的短轴和主轴尺寸分别为0.51 nm×0.55 nm和0.54 nm×0.56 nm),与之相比Beta的12环通道结构(0.65 nm×0.56 nm和0.75 nm×0.57 nm)具有更大的孔径[43]。根据动力学直径(0.62 nm),MTBE分子更易进入Beta的孔径中,而与ZSM-5孔径的相容度不高。Yazaydin等[44]的实验证明Beta的所有孔径都足以容纳MTBE分子。Centi等[45]的实验表明对于具有相同SiO2/Al2O3的两种沸石,Beta对于MTBE的吸附程度更高。因此可以推论,Beta较大的孔径尺寸更有利于容纳MTBE分子。尤其当水中MTBE浓度较高时,在较高的负载量下孔隙体积对吸附能力的影响增强,即孔径较大的沸石更具优越性[24]。如表2所示,与材料疏水性和孔径尺寸相比,比表面积变化对MTBE的吸附过程影响较小。因此,研究沸石的吸附亲和力及孔径尺寸对于进一步评估其对污染物质的降解效率是非常重要的。
另一方面,Beta对H2O2的分解和MTBE的降解具有相对较低的催化活性。与ZSM-5(3.7×10−3 L·g−1·min−1)相比,Beta(3.0×10−5 L·g−1·min−1)减少了两个数量级。H2O2的分解率较低导致生成的处于稳定状态的OH·的浓度成比例降低[38]。造成其催化活性差异的原因如下:如表2所示,其一是铁含量的不同,另外铁的形态差异亦有明显影响。实验证明,可通过水热合成[47-48]或离子交换[49]将过渡金属(主要是铁)引入沸石中。根据合成途径及铁含量的不同,引入沸石中的铁以不同的形态存在:从离子交换位点分离出的铁离子;骨架结构中的铁原子;沸石表面的氧化铁颗粒等[50]。其中,原始材料中的铁大多以骨架铁的形态存在,在氧化过程中的反应活性很小;附着在沸石上的铁离子大多具有较高的氧化活性,能充分发挥催化作用[41, 51-52];而氧化铁颗粒可以促进H2O2分解以形成更多的OH·[46]。为了进一步阐明不同种类含铁沸石在催化氧化过程中的作用,需要更广泛的研究。
-
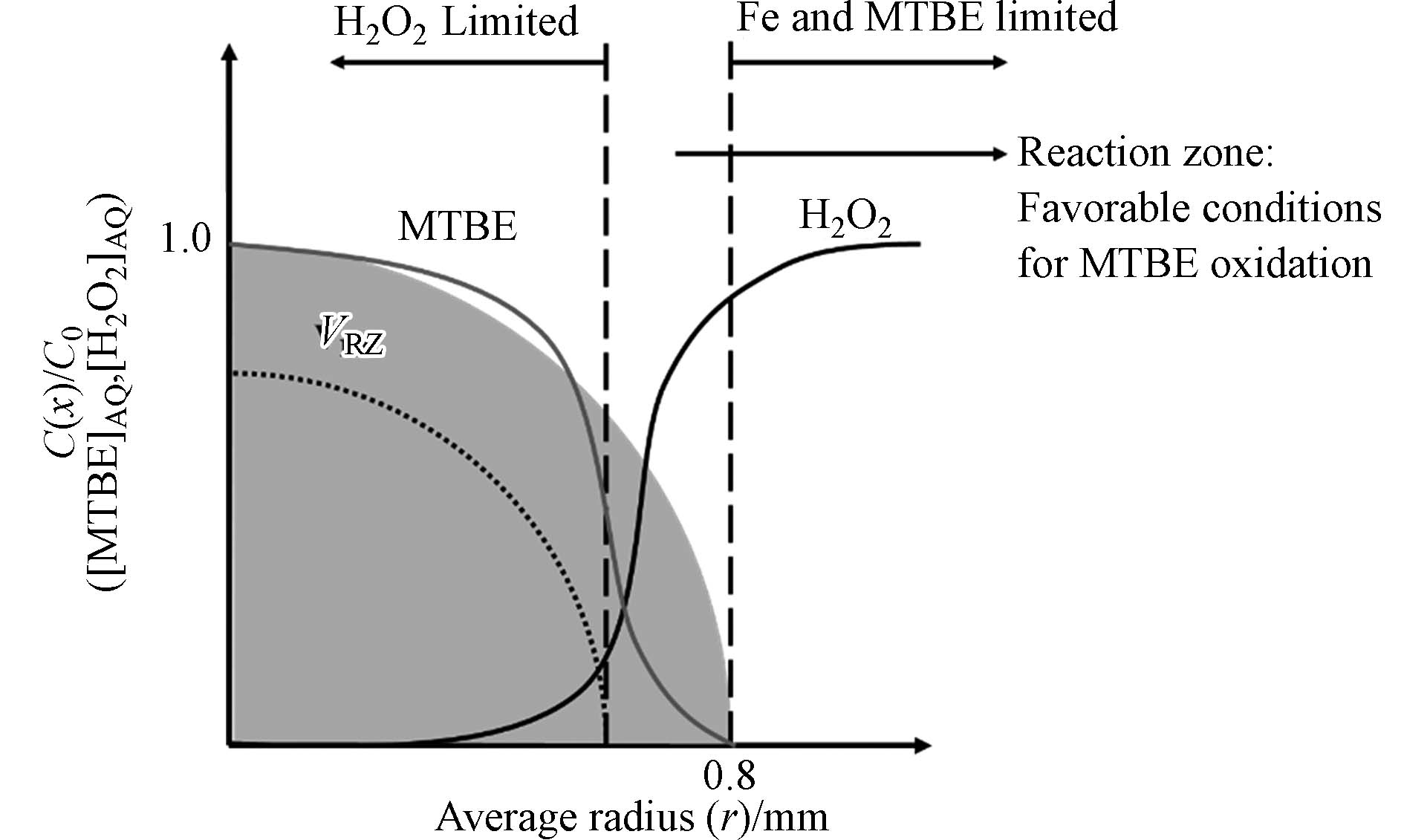
活性炭(Activated Carbon, AC)廉价易得,可用于处理较大范围的污染,因而针对AC的研究更加全面,工艺设备可靠。该方法涉及H2O2与Fe在AC上形成OH·的分解反应、吸附固定在炭粒中MTBE的解吸扩散过程及MTBE在反应区域内的氧化降解反应。反应主要发生在AC上而不是溶液中[53]。其中,MTBE从AC内部的解吸过程涉及以下步骤:(i)从固相解吸到液相;(ii)孔隙内部的传递;(iii)扩散穿过颗粒边缘的静态薄膜;(iv)进入到溶液中[54]。与此同时,H2O2从溶液向炭粒内部迁移并与吸附固定在AC上的铁反应[53]。如图4所示,在AC中存在一个H2O2、Fe及MTBE均能以充分浓度共存的反应区域,以完成MTBE的氧化降解与AC的再生反应[55]。反应区域的大小取决于铁的渗透、H2O2的扩散及颗粒内部MTBE的解吸和运输距离。由于铁的渗透程度有限,加之H2O2的快速反应使H2O2的扩散距离受限,使反应区域主要存在于颗粒的外围。研究表明,MTBE的降解率与AC粒径之间呈线性负相关关系。在粒径较小的AC中H2O2和MTBE所需迁移距离较小,反应区域所占比例相对较高,且粒径的差异并不会改变其物化性质[56]。基于该分析可推断,反应区体积与总体积之比(VRZ/VT)与粒径呈反比,即小颗粒有更大的处理潜力。
AC的零电荷点(pHPZC)指颗粒的表面电荷呈中性,所含正电荷数与负电荷数大致相等的pH值[58-59]。若溶液pH<pHPZC,AC表面正电离子与溶液中铁离子之间的同性相斥力会阻止铁向颗粒内部迁移。因此在实验中需要采用酸性铁试剂对AC修饰改性[60-61],以增加AC的阳离子交换容量,保证可溶铁离子的有效渗透[62-63]。其作用原理如下:通过引入各类酸根离子氧化炭粒表面[64]以降低pHPZC,使溶液pH>pHPZC。此时,炭粒表面被带有负电荷的酸根离子覆盖,能通过异性相吸作用吸附溶液中的铁离子,有利于铁离子的迁移扩散[65]。Tseng等[66]在实验过程中通过酸处理将pHPZC从5.0降到3.0—3.4,在pH≥pHPZC的条件下提高了对MTBE的氧化程度,即预处理过程是有必要的。
对于预处理中采用的酸性试剂,如表3所示,不同价态的铁试剂会有不同的效果。由于Fe3+更不稳定,其溶解度与pH成反比,反应后溶液pH升高(pH≥3),Fe3+会在颗粒表面絮凝沉淀[67]。使铁主要积累于颗粒边缘,甚至堆积的固态沉淀物会堵塞炭粒的孔隙,导致颗粒的比表面积减少[68],不利于Fe3+的扩散,反应区域较小。而Fe2+的溶解度较高,与颗粒表面的反应较弱,可渗透到炭粒中心,反应区域较大,并可减少对MTBE吸附的干扰[69]。不同种类的酸根离子亦会有不同的影响。Fe3+可与
${\rm{SO}}_4^{2 - } $ 、Cl−形成多种配合物(FeCl2+、${\rm{FeSO}}_4^ + $ ),使${\rm{SO}}_4^{2 - } $ 、Cl−被固定在炭粒上,被固定的硫不会干扰反应。目前还没有发现Fe3+与${\rm{NO}}_3^ - $ 、${\rm{ClO}}_4^ - $ 之间的络合反应,反应后将继续存在于溶液中[70]。然而,实验中并不期望在溶液中残留有${\rm{NO}}_3^ - $ 、${\rm{ClO}}_4^ - $ 。EPA规定的水环境中硝酸盐和高氯酸盐可存在的最高浓度分别为10 mg·L−1[71]及15 μg·L−1[72]。因此,若使用Fe(NO3)3或Fe(ClO4)3溶液则均需要额外采取适当的措施以消除溶液中残留的酸根离子。这不仅增加了处理难度亦产生了额外的处理费用,所以在实验过程中不建议采用。除了上文所述吸附材料的种类、粒径[57]及不同的酸性铁试剂[69]之外,还有诸多因素会影响MTBE的降解效果,如H2O2的浓度[53]、温度[55]等。对于H2O2应控制其浓度在合适的范围。一方面,尽管H2O2和OH·之间的反应速率常数较低(2.7×107 L·mol−1s−1),高浓度的H2O2本身亦会和OH·反应,消耗自由基的数量[25]。即在一定浓度范围内氧化效率会随H2O2浓度的增加而降低[53]。另一方面,高浓度的H2O2在AC中的扩散范围更大,与铁接触反应的机会更多,可持续产生OH·以提高氧化反应的速率和程度[73]。因此,应根据处理目标选择合适的浓度,例如,若需要获得最短的反应时间则应选择高浓度的试剂。但若考虑到成本及效率问题,较低浓度的试剂则更为经济。此外,温度对处理效果亦有显著影响。Kan等[55]的实验结果显示,在21—27℃下测得MTBE的去除率较低(62%—80%),而将温度升至50 ℃下测得的去除率则可维持在较高水平(92%—97%)。他们推测这是由于高温有利于物质的迁移,使各反应物均匀分散,扩大了反应区域,促进了对MTBE的氧化降解。
该联合修复技术的优势在于首先,目标污染物在反应位点附近的吸附富集增加了其与OH·之间发生碰撞及反应的概率,提高了反应效率;其次,非均相反应的优点在于可以在中性条件下工作,具有回收铁资源的可能性并避免了含铁污泥的形成,可用于固定床反应器;此外,该方法可通过氧化反应对吸附剂进行再生处理,降解吸附固定在吸附剂上的污染物。可有效延长吸附剂的使用寿命,降低运行成本,解决吸附法固有的废吸附剂污染问题。
其不足之处在于,该技术主要利用OH·氧化降解被吸附而存在于颗粒表面或孔径内的污染物。然而反应生成的OH·大多存在于液相中,由于OH·反应速度极快、存在时间太短而较难扩散进入到吸附材料的孔径中,因此反应区域有限。另外,为了保持并提高非均相反应的活性,需要提供充足的H2O2试剂,并及时通过离子交换引入以补充材料中的铁。
-
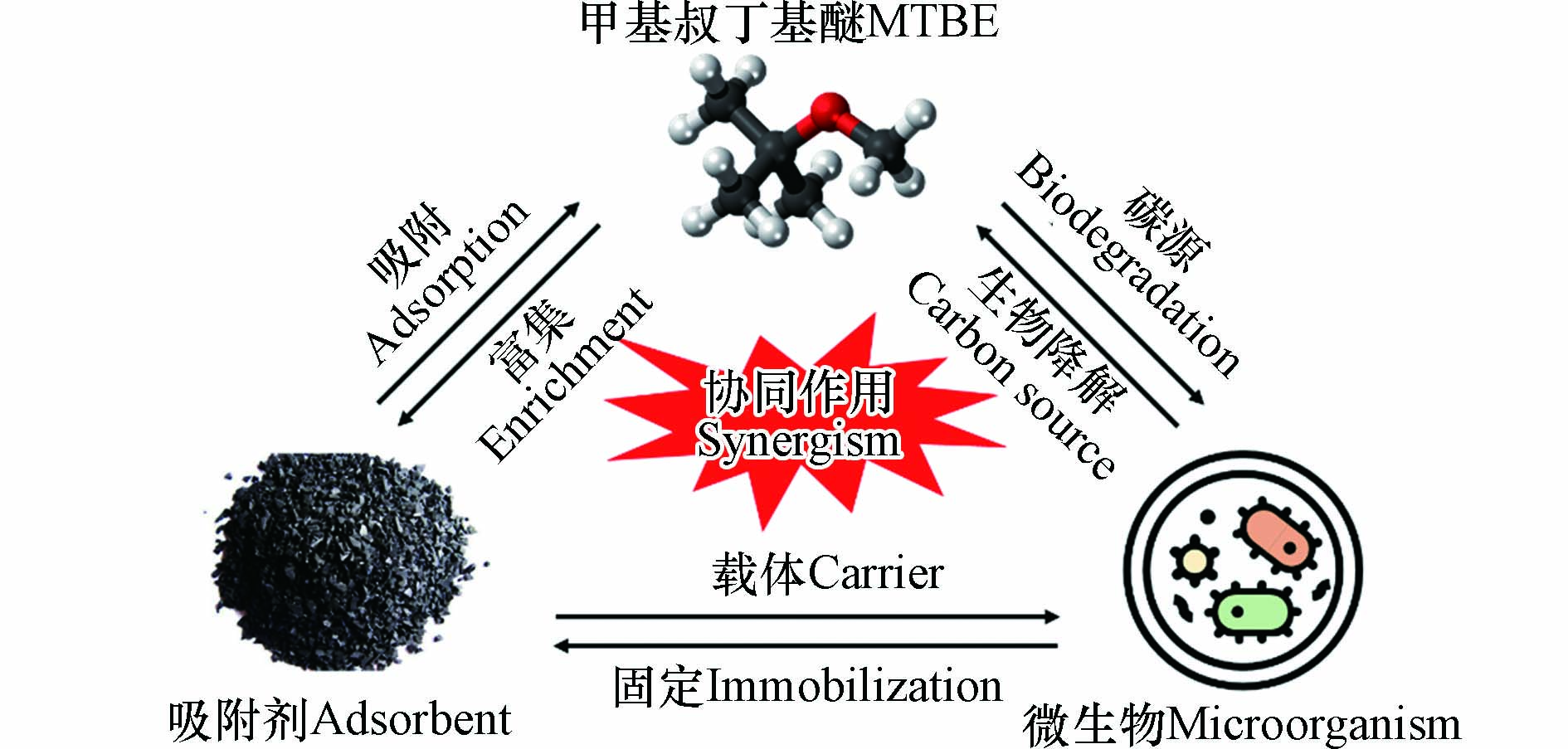
由于吸附技术会受到吸附容量的限制,并且会在达到饱和吸附量后产生二次废物,而微生物可在其生长过程中降解污染物[74-75],因此,可将吸附与生物修复技术联用。即微生物以吸附材料为载体生长,在吸附的同时利用微生物将污染物完全降解为CO2和H2O或其他毒性更小的物质[76-77],其机理示意图如图5所示。一方面,载体为降解菌提供营养物质;另一方面,菌体可通过降解载体吸附的部分污染物而实现对载体材料的再生。Alfonso-Gordillo等[78]将降解MTBE的混合菌群固定于火山岩颗粒表面,构成上流式填充床生物膜反应器,去除速率达到了128.3 mg·L−1·h−1,降解效率超过90%,该反应器对MTBE的持续降解非常有效。Vainberg等[79]以AC为载体吸附剂固定混合菌群,建立流化床反应器,实现了将MTBE的浓度从进水的7.3 mg·L−降至20 μg·L−1。Lyew等[80]比较了MTBE降解菌Mycobterium austroafricanum IFP分别在泥炭、火山灰、颗粒活性炭、珍珠岩、浮石这五种载体上的负载情况。通过分析IFP对MTBE的降解情况,确定珍珠岩是最利于IFP生长及MTBE降解的载体。同时比较了不同材料对MTBE的吸附能力,证实AC的吸附能力最好。Pongkua等[81]的实验亦表明,微生物可长期附着生长在吸附材料上,即该联合技术降解MTBE是可行的。
目前已经发现并分离出了许多可以降解MTBE的微生物,包括Methylibium petroleiphilum PM1 [82]、Hydrogenophaga flava ENV735 [18]、Aquincola tertiaricarbonis L108 [83]等。在该方法中,载体材料是微生物生长的环境,合适的载体能提高降解反应的速率。影响微生物在材料上负载的因素较多,如比表面积、孔隙率、孔径结构、表面活性基团等。通常,有较大比表面积和孔径体积,呈弱碱性的载体负载的生物量较大[84],粗糙的表面结构能增加与MTBE分子各官能团接触反应的机会[85- 86]。对于载体的形状,粒状材料中含有更多的水分,不利于MTBE的扩散,因而对MTBE的吸附容量较低。但在长期运行的情况下,有较高的水分含量更适合降解菌生长[87-88]。并且粒状材料更易于与葡萄糖浆等混合,能够为细菌的生长提供足够的碳源和能源,以保持降解菌的活性。因此粒状材料则更适合在处理时间较长的生物系统中与微生物联用[89-90]。
为了长期提高MTBE的去除效率,在微生物的选择上可优先考虑从被汽油污染的环境中分离出的微生物。因为此类微生物对MTBE的毒性有较好的耐受效果,能在长期接触MTBE的情况下保持良好的生存状况[91-92]。此外,还可在系统中添加新的营养源,创造合适的环境以供微生物生长[93]。或通过添加微生物悬浮液,以接种微生物的方式维持系统中活性降解菌的数量[94-95]。且实验表明中性环境对微生物的生长及污染物的去除更有利[3]。
对于载体上负载微生物的数量,并不是越多去除效率越高。研究表明,若固定于载体表面的降解菌数量过多,在一定程度上会占据材料的吸附位点,影响MTBE进入材料的传质过程。Wang等[96]的实验中就出现过此类现象。一方面,固定生长在材料表面的微生物本身能够充分降解MTBE。另一方面,其会与MTBE竞争材料本身的吸附作用,并且若负载的微生物过多还会存在因营养物质不足而影响降解效率的情况[97]。即需要根据具体情况比较降解菌增加的去除效率与载体损失的吸附效率之间的关系[98]。因此,在实验过程中要合理控制固定菌株的数量。大量实验证明,在微生物数量适当的情况下,与单独应用相比,该联合处理技术能明显提高对MTBE的去除率。然而,目前关于联合作用情况下这两种方法各自所占分配比例的研究较少[99]。文献中普遍认同这两类处理技术是协同作用,均对污染物的去除有足够的贡献。并不能简单的认为其中一种方法占主导作用,也不只是两种作用的简单合并[98]。
该方法的不足之处在于,在长期实验后降解效率会有所下降。其可能的原因在于首先,MTBE具有毒性,与微生物直接接触并长期暴露会使其中毒,对微生物的富集性和多样性产生负面影响。此外,由于微生物附着生长在吸附材料上,若载体不能提供充足的营养物质,则会影响微生物的活性。因此有必要通过在吸附材料中混合葡萄糖浆等步骤以增加降解菌生长所需要的营养。
综上,该联合处理技术能有效降解MTBE,并能在更长的时间内保持较高的降解效率。且与游离菌株相比,将微生物负载于吸附材料上具有多种优势。如易于固液分离、提高了菌株抗冲击负荷的能力并使其维持在高浓度水平。此外,该方法不仅能延长载体吸附剂的使用寿命,降低运行成本,还可避免产生二次污染,增加废物二次利用的价值,具有生态友好性。
-
高级氧化法的成本较高加之氧化剂的寿命有限,通常仅为几小时到几天,其有效性会受到限制[100-101]。生物降解技术会被环境中污染物浓度、温度等因素影响。为了克服两种技术单独应用时固有的局限性,可将其联合应用。据报道,高级氧化-生物降解联合修复技术在原位修复受苯、甲苯、乙苯、二甲苯、MTBE、TBA污染的地下水的诸多案例中已经获得了成功应用[102-104]。
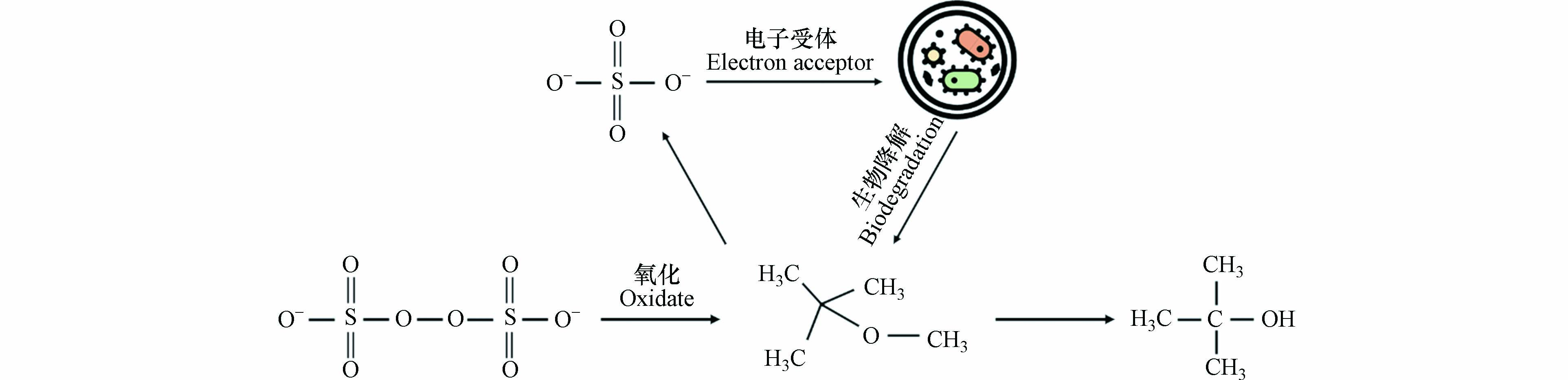
例如,过硫酸盐是一种常用的化学氧化剂,适用于多种污染物并可在水环境中保持较长时间的稳定性[105-106]。在过硫酸盐氧化MTBE的反应过程中其本身被还原为硫酸盐,生成的硫酸盐可作为硫酸盐还原细菌的电子受体。且已有实验证明,该细菌可降解包括MTBE在内的多种有机化合物[107],是一种有效的联合处理方法,其作用机理示意图如图6所示。
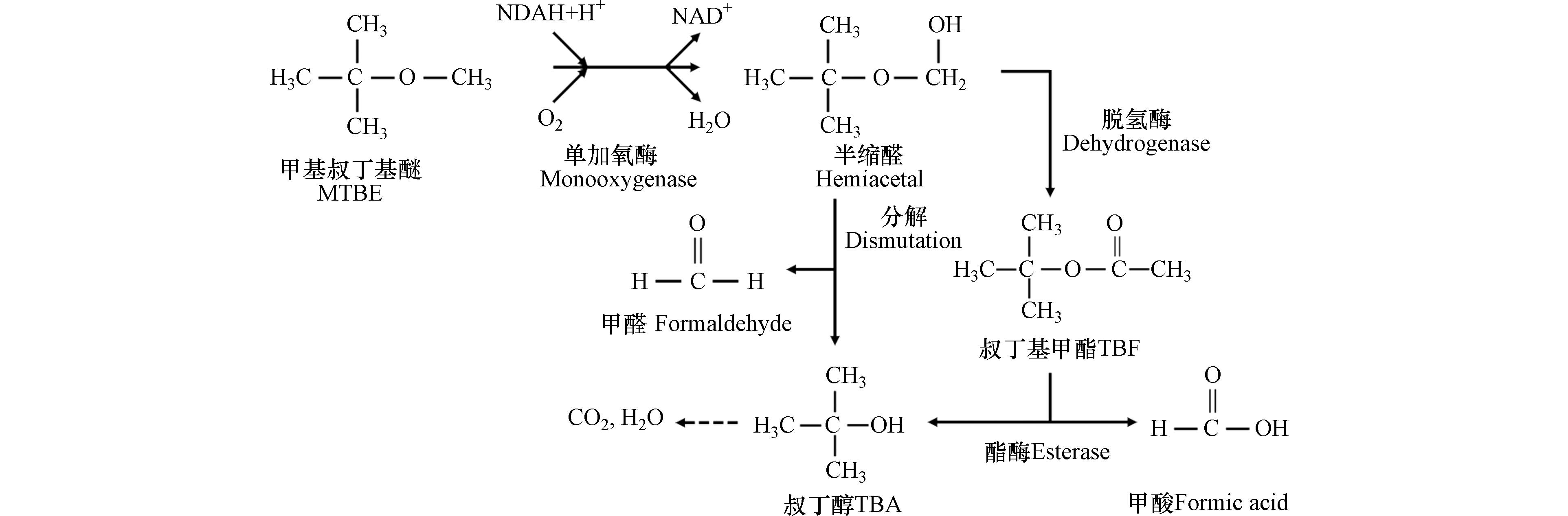
McGregor等[108]的实验表明,MTBE和TBA的浓度随时间变化的趋势不同。在同一时间段内,随MTBE浓度的降低,TBA的浓度增加,表明MTBE结构中的醚键被降解形成TBA。当MTBE的浓度低于一定范围后,TBA的浓度开始呈现下降趋势。证明该联合处理技术可进一步降解反应产生的中间产物,不会残余有毒或难降解的副产物。在该过程中,首先进行单加氧酶所催化的羟化反应,而后以半缩醛形式存在的产物叔丁氧基甲醇或迅速分解为TBA和HCHO,或经由TBF水解为TBA和HCOOH。其可能的降解途径如图7所示[109],随后TBA经过一系列反应最终被完全降解。
其局限性在于,若环境中有
${\rm{CO}}_3^{2-} $ 、Cl−、Br−等同时存在会与目标污染物竞争氧化剂的作用,消耗氧化剂的数量。同时,若缺少足够量的${\rm{SO}}_4^{2-} $ 则会限制微生物的活性,不利于MTBE的降解。其优点在于可通过协同效应弥补两种技术独立应用时的缺点,在提高降解速率的同时避免产生二次污染。且该方法的适用范围广泛,可应用于包括盐水层在内的各类水环境中处理多种有机污染物。综上,该联合修复技术可高效降解MTBE,具有广泛的应用前景。 -
本文对3类典型的去除环境中MTBE污染的联合处理技术进行了总结。回顾本文可以了解到在这3类方法中“吸附-高级氧化联合处理技术”的降解效率高,可适用于处理较大范围的污染;“吸附-生物降解联合处理技术”的成本较低,对环境更加友好,不会产生二次污染;“高级氧化-生物降解联合处理技术”需要合适的氧化剂与相应的菌种,不适合处理成分复杂的样品。根据MTBE的污染现状,有必要完善监控系统,进一步加强对MTBE的检测力度。并根据污染地的环境差异及预算、处理要求等因素,选择适当的方法修复环境污染,减少对人体健康产生的危害。
纳米材料在水处理等诸多领域均有广泛的应用前景,例如,碳纳米管(Carbon Nanotubes, CNTs)或可作为一种良好的吸附材料用于有机污染物的吸附,或可用作稳定的载体材料。此外,常见的纳米材料还包括纳滤膜、纳米零价铁、膨润土等。在未来的发展中可将其与降解MTBE的联合处理技术结合,目前,对不同纳米材料在降解MTBE污染方面的应用亟待进行详细的研究。另外一种防止MTBE污染环境的方法是寻找一种环境风险小、不会对人或动物健康构成威胁的、应用前景显著的替代产品。如以乙基叔丁基醚(Ethyl tert-butyl ether, ETBE)或乙醇等清洁燃料作为汽油添加剂,从源头上减少MTBE的用量。
典型联合处理技术去除甲基叔丁基醚污染的研究进展
Research progress on typical combined treatment technologies for removal of methyl tert-butyl ether
-
摘要: 甲基叔丁基醚(MTBE)作为汽油添加剂的广泛使用造成了世界范围内的环境污染。考虑到其毒性及高水溶性、抗降解性等,会在环境中造成持久性污染。因此,开发高效便捷的处理技术已刻不容缓。单一的降解方法存在不同的局限性,而将不同处理技术联合应用,取长补短,能够克服各方法单独应用时固有的问题,促进协同效应,提高甲基叔丁基醚的降解率。基于物理吸附、高级氧化、生物降解技术各自的优点,文章总结了近年来应用上述3种方法的联合处理技术的研究进展,阐述了处理效果和作用机理,同时对未来的研究方向提出展望。Abstract: The extensive use of methyl tert-butyl ether (MTBE) as a gasoline additive has resulted in widespread distribution of this compound in the environment. Due to its potential toxicity, high solubility and recalcitrance to degradation, the distribution of MTBE in the environment may lead to persistent pollution. Thus, the development of technology to eliminate MTBE contamination has become a priority. The existing single treatment technique has various limitations. However, the combined application of different processes could avoid the limitations and promote synergistic effects of single technique, resulting in the increasing of MTBE degrading rates. Based on the above description, this article summarizes the recently development and mechanism of combined techniques, including physical adsorption method, advanced oxidation method, and biodegradation method. The prospects for future research have also been proposed.
-
Key words:
- MTBE /
- combined treatment technology /
- degradation of pollutants
-

-
表 1 各联合修复技术的优缺点
Table 1. The advantages and disadvantages of each combined technology
方法
Method优点
Advantage缺点
Disadvantage沸石吸附-芬顿氧化 提高了降解效率,可在中性环境中工作,具有回收铁资源的可能性并可避免形成含铁污泥.沸石的化学性质稳定,可选择性吸附有机物,应用范围广泛. 需要在多种因素共存的条件下进行,反应区域有限;载体吸附剂的再生效率不稳定;
对操作条件的要求较为严格.活性炭吸附-芬顿氧化 成本较低,适用于处理较大范围的污染,工艺设备可靠,且AC的负载的能力较高,吸附性能较好. 吸附-生物降解 能在更长时间内保持较高的降解效率;易于固液分离,菌株密度高,提高了菌株抗冲击负荷的能力;延长载体的使用寿命,降低运行成本;避免二次污染,具有生态友好性. 长期运行后其降解效率有所下降,需要选择合适的吸附材料并提供充足的营养物质 高级氧化-生物降解 提高了降解速率;避免产生二次污染,适用范围广泛,可处理多种有机污染物. 需要充足的氧化剂,不适合处理成分复杂的样品. 表 2 沸石的特性
Table 2. The characteristics of zeolite
沸石
Zeolite最大自由孔径
Maximum free pore diameter/nm铁含量/%
Fe content(wt)BET表面积/ (m 2 ·g −1)
BET surface area粒径/μm
Particle sizeSiO2/Al2O3 来源
ReferenceZSM5 0.56 N.D.c 330 1.0—17a 683 [46] ZSM5 0.56 0.37 265 63—200b 400 [46] ZSM5 0.56 0.03 385 4.4—7.1a 236 [46] Fe-ZSM5 0.56 2.2 370 6.4—12a 26 [46] H-ZSM5 — — 450 1—3 25 [45] H-ZSM5 — — 425 1—3 80 [45] Na-ZSM5 — — 430 1—3 25 [45] Beta 0.75 N.D. 580 5.6—15a 200 [46] Fe-Beta 0.75 1.3 600 1.0—2.1a 35 [46] Fe-Beta 0.75 3.1 600 250—630b 25 [46] H-Beta — — 680 0.2—1.0 25 [45] H-mordenite — — 480 0.2—1.2 15 [45] H-faujasite — — 780 1—3 30 [45] a通过激光衍射分析确定的下限d50—上限d90.b筛分的粒状物料.c未检出.
a Lower limit d50—upper limit d90 determined by laser diffraction analysis. b Sieved fractions of pelletized materials. c Not detected.表 3 不同铁溶液对AC修饰改性的结果
Table 3. Results of various Fe amendment to the AC
铁类型
Fe type[Fe]initial/
(g·L−1)a[Fe]add/
(mg·kg−1)b[Fe]AC/
(mg·kg−1)[Fe]final/
(mg·L−1)epH
(Post oxidation)保留率/%
Retention ratef来源
Reference硫酸铁 低 1.25 3640 5480 c — 3.1 98.9 [69] 中 2.50 6900 8740 c — 3.0 96.8 [69] 高 3.75 8750 10590 c — 2.8 90.7 [69] 氯化铁 低 1.25 3310 5150 c — 3.1 95.2 [69] 中 2.50 5780 7620 c — 3.0 90.3 [69] 高 3.75 6960 8800 c — 2.7 83.8 [69] 硝酸铁 低 1.25 3440 5180 c — 3.2 96.6 [69] 中 2.50 6850 8690 c — 2.9 96.5 [69] 高 3.75 7850 9690 c — 2.7 87.2 [69] 硫酸亚铁 低 1.25 3730 5570 c — 3.1 99.9 [69] 低 2.50 7450 9290 c — 3.0 99.9 [69] 中 3.75 11060 12900 c — 2.9 99.5 [69] 中 4.40 — 12630d 97 — 98.0 [53] 中 5.50 — 15760d 270 — 95.0 [53] 高 6.60 — 16900d 730 — 89.0 [53] 高 8.20 — 18520d 1590 — 81.0 [53] a初始溶液中的铁浓度.b引入到AC中的铁浓度.
c[Fe]AC:AC上固定的铁=引入的铁+环境中的铁([Fe]AC=1840 mg/kg(n=14);95%的置信区间为1500—2170 mg/kg).
d[Fe]AC:AC上固定的铁=引入的铁+环境中的铁([Fe]AC=1020 mg/kg(n=3).e反应后溶液中的铁浓度.
f保留百分率=(([Fe]initial-[Fe]final)/[Fe]initial)×100%.
a Fe concentration in initial aqueous. b Amended Fe concentration immobilized in the AC.
c [Fe]AC: Total Fe on AC=Fe amended+ average background([Fe]AC=1840 mg/kg(n=14);95% confidence interval,1500–2170 mg/kg).
d [Fe]AC: Total Fe on AC=Fe amended+ average background([Fe]AC=1020 mg/kg(n=3). e Fe concentration in solution after reaction.
f Values in parentheses represent percent retention= (([Fe]initial-[Fe]final)/[Fe]initial) ×100%. -
[1] LINARES M R, BRUGUERA S L, BARCELó D. Analysis, occurrence and fate of MTBE in the aquatic environment over the past decade [J]. Trac Trends in Analytical Chemistry, 2006, 25(10): 1016-1029. doi: 10.1016/j.trac.2006.06.011 [2] WILLIAMS M. The merck index: An encyclopedia of chemicals, drugs, and biologicals, 15th edition edited [J]. Drug Development Research, 2013, 74(5): 339-339. doi: 10.1002/ddr.21085 [3] RUSSO A V, LOBO D N D, JACOBO S E. Removal of MTBE in columns filled with modified natural zeolites [J]. Procedia Materials Science, 2015, 8: 375-382. doi: 10.1016/j.mspro.2015.04.087 [4] FRANCISCO FACETTI J, NUNEZ R, GOMEZ C L, et al. Methyl tert-butyl ether (MtBE) in deep wells of the Patino Aquifer, Paraguay: A preliminary characterization [J]. The Science of the Total Environment, 2019, 647: 1640-1650. doi: 10.1016/j.scitotenv.2018.08.062 [5] LEVCHUK I, BHATNAGAR A, SILLANPää M. Overview of technologies for removal of methyl tert-butyl ether (MTBE) from water [J]. Science of The Total Environment, 2014, 476/477: 415-433. doi: 10.1016/j.scitotenv.2014.01.037 [6] ORGANIZATION W H. Guidelines for drinking-water quality: fourth edition[R]. World Health Organization, 2011. [7] AGENCY U S E P. 2012 Edition of the Drinking Water Standards and Health Advisories[R]. 2012. [8] GARRETT P, MOREAU M. MTBE as a ground water contaminant [J]. Prevention, Detection and Restoration, 1986: 227-238. [9] CHEN C S, HSEU Y C, LIANG S H, et al. Assessment of genotoxicity of methyl-tert-butyl ether, benzene, toluene, ethylbenzene, and xylene to human lymphocytes using comet assay [J]. Journal of Hazardous Materials, 2008, 153(1): 351-356. [10] EPA U. Oxyfuels Information Needs[R]. 1996. http://cfpub.epa.gov/ncea/cfm/recordisplay.cfm?deid=29060. [11] SAHAY N M T, MARCHIONNA M. Low-cost conversion of MTBE units to make alternative gasoline blending components [J]. World Refining, 2002, 12(7): 34-70. [12] 《中国能源》编辑部. 《关于扩大生物燃料乙醇生产和推广使用车用乙醇汽油的实施方案》印发 [J]. 中国能源, 2017, 39(9): 1. Editorial Department Of China Energy. Implementation plan for expanding the production of bio fuel ethanol and popularizing the use of ethanol gasoline for vehicles [J]. China Energy, 2017, 39(9): 1(in Chinese).
[13] VIGNOLA R, BAGATIN R, D’AURIS A D F, et al. Zeolites in a permeable reactive barrier (PRB): One year of field experience in a refinery groundwater—Part 1: The performances [J]. Chemical Engineering Journal, 2011, 178: 204-209. doi: 10.1016/j.cej.2011.10.050 [14] VIGNOLA R, COVA U, FABIANI F, et al. Remediation of hydrocarbon contaminants in groundwater using specific zeolites in full-scale pump&treat and demonstrative Permeable barrier tests [J]. Studies in Surface Science & Catalysis, 2008, 174(8): 573-576. [15] BURBANO A A, DIONYSIOU D D, SUIDAN M T, et al. Oxidation kinetics and effect of pH on the degradation of MTBE with Fenton reagent [J]. Water Research, 2005, 39(1): .107-118. doi: 10.1016/j.watres.2004.09.008 [16] SALANITRO J P, DIAZ L A, WILLIAMS M P, et al. Isolation of a bacteria culture that degrades methyl t-butyl ether [J]. Applied & Environmental Microbiology, 1995, 61(1): 406. [17] KANE S R, BELLER H R, LEGLER T C, et al. Aerobic biodegradation of methyl tert-butyl ether by aquifer bacteria from leaking underground storage tank sites [J]. Applied & Environmental Microbiology, 2002, 67(12): 5824-5829. [18] HATZINGER P B, MCCLAY K, VAINBERG S, et al. Biodegradation of methyl tert-butyl ether by a pure bacterial culture [J]. Applied & Environmental Microbiology, 2001, 67(12): 5601-5607. [19] BURGHOFF B, MARQUES J S, LANKVELT B M V, et al. Solvent impregnated resins for MTBE removal from aqueous environments [J]. Reactive & Functional Polymers, 2010, 70(1): 41-47. [20] KELLER A, SANDALL O, RINKER R, et al. Cost and performance evaluation of treatment technologies for MTBE-contaminated water[J]. 1998. [21] MOHEBALI S. Degradation of methyl t-butyl ether (MTBE) by photochemical process in nanocrystalline TiO2 slurry: Mechanism, by-products and carbonate ion effect [J]. Journal of Environmental Chemical Engineering, 2013, 1(4): 1070-1078. doi: 10.1016/j.jece.2013.08.022 [22] KIM D K, O’SHEA K E, COOPER W J. Mechanistic considerations for the degradation of methyl tert-butyl ether (MTBE) by sonolysis: Effect of argon vs. oxygen saturated solutions [J]. Ultrasonics Sonochemistry, 2012, 19(4): 959-968. doi: 10.1016/j.ultsonch.2011.12.003 [23] ANDERSON, MICHAEL A. Removal of MTBE and other organic contaminants from water by sorption to high silica zeolites [J]. Environmental Science & Technology, 2000, 34(4): 725-727. [24] ERDEM-SENATALAR A, BERGENDAHL J A, GIAYA A, et al. Adsorption of methyl tertiary butyl ether on hydrophobic molecular sieves [J]. Environmental Engineering Science, 2004, 21(6): 722-729. doi: 10.1089/ees.2004.21.722 [25] BUXTON G V, GREENSTOCK C L, HELMAN W P, et al. Critical review of rate constants for reactions of hydrated electronschemical kinetic data base for combustion chemistry. Part 3: Propane [J]. Journal of Physical & Chemical Reference Data, 1988, 17(2): 513-886. [26] BURBANO A A, DIONYSIOU D D, SUIDAN M T. Effect of oxidant-to-substrate ratios on the degradation of MTBE with Fenton reagent [J]. Water Research, 2008, 42(12): 3225-3239. doi: 10.1016/j.watres.2008.04.004 [27] RUPPERT G, BAUER R, HEISLER G. The photo-Fenton reaction-an effective photochemical wastewater treatment process [J]. Journal of Photochemistry & Photobiology A Chemistry, 1993, 73(1): 75-78. [28] 刘勇弟, 徐寿昌. 紫外-Fenton试剂的作用机理及在废水处理中的应用 [J]. 环境化学, 1991, 34(4): 302-306. LIU Y D, XU S C. Study on the reaction mechanism of UV-Fenton reagent and its application to wastewater treatment [J]. Environmental Chemistry, 1991, 34(4): 302-306(in Chinese).
[29] ZHOU M, TAN Q, QIAN W, et al. Degradation of organics in reverse osmosis concentrate by electro-Fenton process [J]. Journal of Hazardous Materials, 2012, 215/216(15): 287-293. [30] OTURAN M A, AARON J J. Advanced oxidation processes in water/wastewater treatment: Principles and applications. A review [J]. Critical Reviews in Environmental Ence & Technology, 2014, 44(23): 2577-2641. [31] VORONTSOV A V. Advancing Fenton and photo-Fenton water treatment through the catalyst design [J]. Journal of Hazardous Materials, 2019, 372(15): 103-112. [32] 张德莉, 黄应平, 罗光富, 等. Fenton及Photo-Fenton反应研究进展 [J]. 环境化学, 2006, 25(2): 121-127. doi: 10.3321/j.issn:0254-6108.2006.02.001 ZHANG D L, HUANG Y P, LUO G F, et al. Research progress of Fenton and photo-Fenton reaction [J]. Environmental Chemistry, 2006, 25(2): 121-127(in Chinese). doi: 10.3321/j.issn:0254-6108.2006.02.001
[33] SEDLAZECK K P, VOLLPRECHT D, MüLLER P, et al. Decomposition of dissolved organic contaminants by combining a boron-doped diamond electrode, zero-valent iron and ultraviolet radiation [J]. Chemosphere, 2019, 217: 897-904. doi: 10.1016/j.chemosphere.2018.11.043 [34] STEFAN M I, MACK J, BOLTON J R. Degradation pathways during the treatment of methyl tert-butyl ether by the UV/H2O2 process [J]. Environmental Science & Technology, 2000, 34(4): 650-658. [35] KORYABKINA N, BERGENDAHL J A, THOMPSON R W, et al. Adsorption of disinfection byproducts on hydrophobic zeolites with regeneration by advanced oxidation [J]. Microporous & Mesoporous Materials, 2007, 104(1/3): 77-82. [36] WANG S, LI H, XIE S, et al. Physical and chemical regeneration of zeolitic adsorbents for dye removal in wastewater treatment [J]. Chemosphere, 2006, 65(1): 82-87. doi: 10.1016/j.chemosphere.2006.02.043 [37] ABU-LAIL L, BERGENDAHL J A, THOMPSON R W. Adsorption of methyl tertiary butyl ether on granular zeolites: Batch and column studies [J]. Journal of Hazardous Materials, 2010, 178(1/3): 363-369. [38] GONZALEZ-OLMOS R, ROLAND U, TOUFAR H, et al. Fe-zeolites as catalysts for chemical oxidation of MTBE in water with H2O2 [J]. Applied Catalysis B Environmental, 2009, 89(3/4): 356-364. [39] ZHANG Y H, FEI J, SHEN Z T, et al. Kinetic and equilibrium modelling of MTBE (methyl tert-butyl ether) adsorption on ZSM-5 zeolite: Batch and column studies [J]. Journal of Hazardous Materials, 2018, 347: 461-469. doi: 10.1016/j.jhazmat.2018.01.007 [40] ROSSNER A, KNAPPE D R U. MTBE adsorption on alternative adsorbents and packed bed adsorber performance [J]. Water Research, 2008, 42(8/9): 2287-2299. [41] ZECCHINA A, RIVALLAN M, BERLIER G, et al. Structure and nuclearity of active sites in Fe-zeolites: Comparison with iron sites in enzymes and homogeneous catalysts [J]. Physical Chemistry Chemical Physics, 2007, 9(27): 3483-99. doi: 10.1039/b703445h [42] SCHWIDDER M, KUMAR M S, KLEMENTIEV K, et al. Selective reduction of NO with Fe-ZSM-5 catalysts of low Fe content: Ⅰ. Relations between active site structure and catalytic performance [J]. Journal of Catalysis, 2005, 231(2): 314-330. doi: 10.1016/j.jcat.2005.01.031 [43] SZOSTAK R. Handbook of molecular sieves[M]. Van Nostrand Reinhold, 1992. [44] YAZAYDIN A O, THOMPSON R W. Molecular simulation of the adsorption of MTBE in silicalite, mordenite, and zeolite beta [J]. Journal of Physical Chemistry B, 2006, 110(29): 14458-14462. doi: 10.1021/jp061986n [45] CENTI G, GRANDE A, PERATHONER S. Catalytic conversion of MTBE to biodegradable chemicals in contaminated water [J]. Catalysis Today, 2002, 75(1): 69-76. [46] GONZALEZ-OLMOS R, KOPINKE F D, MACKENZIE K, et al. Hydrophobic Fe-zeolites for removal of MTBE from water by combination of adsorption and oxidation [J]. Environmental Science & Technology, 2013, 47(5): 2353-2360. [47] PéREZ-RAM??REZ J, KUMAR M S, BRÜCKNER A. Reduction of N2O with CO over FeMFI zeolites: Influence of the preparation method on the iron species and catalytic behavior [J]. Journal of Catalysis, 2004, 223(1): 13-27. doi: 10.1016/j.jcat.2004.01.007 [48] RIBERA A, ARENDS I W C E, DE VRIES S, et al. Preparation, characterization, and performance of fezsm-5 for the selective oxidation of benzene to phenol with N2O [J]. Journal of Catalysis, 2000, 195(2): 287-297. doi: 10.1006/jcat.2000.2994 [49] PIRNGRUBER G D, ROY P K, PRINS R. On determining the nuclearity of iron sites in Fe-ZSM-5-a critical evaluation [J]. Physical Chemistry Chemical Physics Pccp, 2006, 8(34): 3939-3950. doi: 10.1039/B606205A [50] DELAHAY G, VALADE D, GUZMáN-VARGAS A, et al. Selective catalytic reduction of nitric oxide with ammonia on Fe-ZSM-5 catalysts prepared by different methods [J]. Applied Catalysis B Environmental, 2005, 55(2): 149-155. doi: 10.1016/j.apcatb.2004.07.009 [51] GONZALEZ-OLMOS R, HOLZER F, KOPINKE F D, et al. Indications of the reactive species in a heterogeneous Fenton-like reaction using Fe-containing zeolites [J]. Applied Catalysis A:General, 2011, 398(1/2): 44-53. [52] MELERO J A, CALLEJA G, MARTÍNEZ F, et al. Crystallization mechanism of Fe-MFI from wetness impregnated Fe2O3–SiO2 amorphous xerogels: Role of iron species in Fenton-like processes [J]. Microporous & Mesoporous Materials, 2004, 74(1/3): 11-21. [53] HULING S G, JONES P K, LEE T R. Iron Optimization for Fenton-driven oxidation of MTBE-spent granular activated carbon [J]. Environmental Science & Technology, 2007, 41(11): 4090-4096. [54] CRITTENDEN J, HAND D, ARORA H, et al. Design considerations for GAC treatment of organic chemicals [J]. Journal American Water Works Association - J AMER WATER WORK ASSN, 1987, 79: 74-82. [55] KAN E, HULING S G. Effects of temperature and acidic pre-treatment on fenton-driven oxidation of mtbe-spent granular activated carbon [J]. Environmental Science & Technology, 2009, 43(5): 1493-1499. [56] HILLING S G, KAN E, CALDWELL C, et al. Fenton-driven chemical regeneration of MTBE-spent granular activated carbon - A pilot study [J]. Journal of Hazardous Materials, 2012, 205/206(29): 55-62. [57] HULING S G, KAN E, WINGO C. Fenton-driven regeneration of MTBE-spent granular activated carbon—Effects of particle size and iron amendment procedures [J]. Applied Catalysis B Environmental, 2009, 89(3/4): 651-658. [58] NOH J S, SCHWARZ J A. Effect of HNO3 Treatment on the Surface Acidity of Activated Carbons [J]. Carbon, 1990, 28(5): 675-682. doi: 10.1016/0008-6223(90)90069-B [59] LOPEZ-RAMON M V, STOECKLI F, MORENO-CASTILLA C, et al. On the characterization of acidic and basic surface sites on carbons by various techniques [J]. Carbon, 1999, 37(8): 1215-1221. doi: 10.1016/S0008-6223(98)00317-0 [60] KINOSHITA K. Carbon: Electrochemical and Physicochemical Properties[M]. John Wiley Sons New York Ny, 1988, 3-12. [61] 范延臻, 王宝贞, 王琳, 等. 改性活性炭对有机物的吸附性能 [J]. 环境化学, 2001, 20(5): 444-448. FAN Y Z, WANG B Z, WANG L, et al. Adsorption of organic micropollutants on modified activated carbons [J]. Environmental Chemistry, 2001, 20(5): 444-448(in Chinese).
[62] HULING S G, JONES P K, ELA W P, et al. Fenton-driven chemical regeneration of MTBE-spent GAC [J]. Water Research, 2005, 39(10): 2145-2153. doi: 10.1016/j.watres.2005.03.027 [63] KIM D S. Adsorption characteristics of Fe(Ⅲ) and Fe(Ⅲ)-NTA complex on granular activated carbon [J]. Journal of Hazardous Materials, 2004, 106(1): 45-54. [64] KARANFIL T, KITIS M, KILDUFF J E, et al. Role of granular activated carbon surface chemistry on the adsorption of organic compounds. 2. Natural organic matter [J]. Environmental Science & Technology, 1999, 33(18): 3225-3233. [65] HUANG H H, LU M C, CHEN J N, et al. Catalytic decomposition of hydrogen peroxide and 4-chlorophenol in the presence of modified activated carbons [J]. Chemosphere, 2003. [66] TSENG H-H, WEY M-Y. Effects of acid treatments of activated carbon on its physiochemical structure as a support for copper oxide in DeSO2 reaction catalysts [J]. Chemosphere, 2006, 62(5): 756-766. doi: 10.1016/j.chemosphere.2005.04.077 [67] BOODT M D, HAYES M H B, HERBILLON A. Soil colloids and their associations in aggregates[M]. 1990: Springer US. [68] REED B E, VAUGHAN R, JIANG L. As(Ⅲ), As(Ⅴ), Hg, and Pb removal by Fe-oxide impregnated activated carbon [J]. Journal of Environmental Engineering, 2000, 126(9): 869-873. doi: 10.1061/(ASCE)0733-9372(2000)126:9(869) [69] HIDING S G, HWANG S. Iron amendment and Fenton oxidation of MTBE-spent granular activated carbon [J]. Water Research, 2010, 44(8): 2663-2671. doi: 10.1016/j.watres.2010.01.035 [70] LAAT J D, LE G T, LEGUBE B. A comparative study of the effects of chloride, sulfate and nitrate ions on the rates of decomposition of H_2O_2 and organic compounds by Fe(Ⅱ)/H2O2 and Fe(Ⅲ)/H2O2 [J]. Chemosphere, 2004, 55(5): 715-723. doi: 10.1016/j.chemosphere.2003.11.021 [71] EPA U. Drinking Water Contaminants 2009. http://www.epa.gov/safewater/contaminants/index.html. [72] EPA U. Interim Drinking Water Health Advisory for Perchlorate. 2008. http://www.epa.gov/waterscience/(EPA 822-R-08-025). [73] HWANG S, HULING S G, KO S. Fenton-like degradation of MTBE: Effects of iron counter anion and radical scavengers [J]. Chemosphere, 2010, 78(5): 563-568. doi: 10.1016/j.chemosphere.2009.11.005 [74] LEBRERO R, LóPEZ J C, LEHTINEN I, et al. Exploring the potential of fungi for methane abatement: Performance evaluation of a fungal-bacterial biofilter [J]. Chemosphere, 2016, 144: 97-106. doi: 10.1016/j.chemosphere.2015.08.017 [75] JOYE S, KLEINDIENST S, PEñA-MONTENEGRO T D. SnapShot: Microbial Hydrocarbon Bioremediation [J]. Cell, 2018, 172(6): 1336-1336.el. doi: 10.1016/j.cell.2018.02.059 [76] XU X, ZHOU H, CHEN X, et al. Biodegradation potential of polycyclic aromatic hydrocarbons by immobilized Klebsiella sp. in soil washing effluent [J]. Chemosphere, 2019, 223(MAY): 140-147. [77] GE J, HUANG G, HUANG J, et al. Mechanism and kinetics of organic matter degradation based on particle structure variation during pig manure aerobic composting [J]. Journal of Hazardous Materials, 2015, 292(15): 19-26. [78] ALFONSO-GORDILLO G, FLORES-ORTIZ C M, MORALES-BARRERA L, et al. Biodegradation of methyl tertiary butyl ether (MTBE) by a microbial consortium in a continuous up-flow packed-bed biofilm reactor: kinetic study, metabolite identification and toxicity bioassays [J]. PLOS ONE, 2016, 11(12): e0167494. doi: 10.1371/journal.pone.0167494 [79] VAINBERG S, TOGNA A P, SUTTON P M, et al. Treatment of MTBE-Contaminated Water in Fluidized Bed Bioreactor [J]. Journal of Environmental Engineering, 2002, 128(9): 842-851. doi: 10.1061/(ASCE)0733-9372(2002)128:9(842) [80] LYEW D, GUIOT S R, MONOT F, et al. Comparison of different support materials for their capacity to immobilize Mycobacterium austroafricanum IFP 2012 and to adsorb MtBE [J]. Enzyme & Microbial Technology, 2007, 40(6): 1524-1530. [81] PONGKUA W, DOLPHEN R, THIRAVETYAN P. Bioremediation of gaseous methyl tert-butyl ether by combination of sulfuric acid modified bagasse activated carbon-bone biochar beads and Acinetobacter indicus screened from petroleum contaminated soil [J]. Chemosphere, 2020, 239: 124724. doi: 10.1016/j.chemosphere.2019.124724 [82] NAKATSU C H, HRISTOVA K, HANADA S, et al. Methylibium petrolelphilum gen. nov. , sp nov. , a novel methyl tert-butyl ether-degrading methylotroph of the Betaproteobacteria [J]. International Journal of Systematic & Evolutionary Microbiology, 2006, 56(Pt 5): 983-989. [83] MüLLER R, ROHWERDER T, HARMS H. Degradation of fuel oxygenates and their main intermediates by Aquincola tertiaricarbonis L108 [J]. Microbiology (Reading, England), 2008, 154: 1414-21. doi: 10.1099/mic.0.2007/014159-0 [84] 何延青, 刘俊良, 杨平, 等. 微生物固定化技术与载体结构的研究 [J]. 环境科学, 2004, 25(s1): 103-106. HE Y Q, LIU J L, YANG P, et al. Study on immobilization technology and carrier structure of microorganism [J]. Environmental Science, 2004, 25(s1): 103-106(in Chinese).
[85] YANG S, LI L, XIAO T, et al. Role of surface chemistry in modified ACF (activated carbon fiber)-catalyzed peroxymonosulfate oxidation [J]. Applied Surface Science, 2016, 383(15): 142-150. [86] EL-SHAFEY E I, ALI S N F, AL-BUSAFI S, et al. Preparation and characterization of surface functionalized activated carbons from date palm leaflets and application for methylene blue removal [J]. Journal of Environmental Chemical Engineering, 2016, 4(3): 2713-2724. doi: 10.1016/j.jece.2016.05.015 [87] PONGKUA W, DOLPHEN R, THIRAVETYAN P. Effect of functional groups of biochars and their ash content on gaseous methyl tert-butyl ether removal [J]. Colloids and Surfaces A:Physicochemical and Engineering Aspects, 2018, 558: 531-537. doi: 10.1016/j.colsurfa.2018.09.018 [88] PONGKUA W, DOLPHEN R, THIRAVETYAN P. Removal of gaseous methyl tert-butyl ether using bagasse activated carbon pretreated with chemical agents [J]. Journal of Chemical Technology & Biotechnology, 2019, 94(5): 1551-1558. [89] FU D, SINGH R P, YANG X, et al. Sediment in-situ bioremediation by immobilized microbial activated beads: Pilot-scale study [J]. Journal of Environmental Management, 2018, 226(11): 62-69. [90] LIU S-H, LIN H-H, LAI C-Y, et al. Microbial community in a pilot-scale biotrickling filter with cell-immobilized biochar beads and its performance in treating toluene-contaminated waste gases [J]. International Biodeterioration & Biodegradation, 2019, 144: 104743. [91] GUISADO I M, PURSWANI J, GONZALEZ-LOPEZ J, et al. Physiological and genetic screening methods for the isolation of methyl tert-butyl ether-degrading bacteria for bioremediation purposes [J]. International Biodeterioration & Biodegradation, 2015, 97: 67-74. [92] WOZNIAK-KARCZEWSKA M, LISIECKI P, BIALAS W, et al. Effect of bioaugmentation on long-term biodegradation of diesel/biodiesel blends in soil microcosms [J]. The ence of the Total Environment, 2019, 671(7): 948-958. [93] HAN M F, WANG C, FU Y. Treatment of hydrophobic volatile organic compounds using two-liquid phase biofilters [J]. Science of the Total Environment, 2018, 640-641(11): 1447-1454. [94] JOYE S B, KLEINDIENST S, GILBERT J A, et al. Responses of microbial communities to hydrocarbon exposures [J]. Oceanography, 2016, 29(3): 136-149. doi: 10.5670/oceanog.2016.78 [95] CAICEDO F, ESTRADA J M, SILVA J P, et al. Effect of packing material configuration and liquid recirculation rate on the performance of a biotrickling filter treating VOCs [J]. Journal of Chemical Technology & Biotechnology, 2018, 93(8): 2299-2306. [96] WANG H, HO L, LEWIS D M, et al. Discriminating and assessing adsorption and biodegradation removal mechanisms during granular activated carbon filtration of microcystin toxins [J]. Water Research, 2007, 41(18): 4262-4270. doi: 10.1016/j.watres.2007.05.057 [97] MULLER R H, ROHWERDER T, HARMS H. Carbon conversion efficiency and limits of productive bacterial degradation of methyl tert-butyl ether and related compounds [J]. Applied and Environmental Microbiology, 2007. [98] 章晶晓. 吸附—生物降解协同去除水相中MTBE的研究[D]. 杭州: 浙江工业大学, 2009. ZHANG J X. Removal of MTBE by adsorption cooperated with biodegradation[D]. Hangzhou: Zhejiang University of Technology, 2009 (inChinese).
[99] 孔令宇, 张晓健, 王占生. 生物活性炭内吸附与生物降解协同去除有机污染物 [J]. 环境科学, 2007, 28(4): 777-780. doi: 10.3321/j.issn:0250-3301.2007.04.015 KONG L Y, ZHANG X J, WANG Z S. Removal of organic pollutants by adsorption cooperated with biodegradation in BAC [J]. Environmental Science, 2007, 28(4): 777-780(in Chinese). doi: 10.3321/j.issn:0250-3301.2007.04.015
[100] HULING S, PIVETZ B. In-situ chemical oxidation [J]. Environmental Protection Agency, 2006: 61. [101] SRA K S, THOMSON N R, BARKER J F. Persulfate treatment of dissolved gasoline compounds [J]. Journal of Hazardous, Toxic, and Radioactive Waste, 2013, 17(1): 9-15. doi: 10.1061/(ASCE)HZ.2153-5515.0000143 [102] CUNNINGHAM J A, RAHME H, HOPKINS G D, et al. Enhanced in situ bioremediation of btex-contaminated groundwater by combined injection of nitrate and sulfate [J]. Environmental Science & Technology, 2001, 35(8): 1663-1670. [103] USMAN M, FAURE P, RUBY C, et al. Remediation of PAH-contaminated soils by magnetite catalyzed Fenton-like oxidation [J]. Applied Catalysis B:Environmental, 2012, 117-118: 10-17. doi: 10.1016/j.apcatb.2012.01.007 [104] SHAYAN M, THOMSON N R, ARAVENA R, et al. Integrated plume treatment using persulfate coupled with microbial sulfate reduction [J]. Groundwater Monitoring & Remediation, 2018, 38(4): 45-61. [105] HUANG K C, ZHAO Z, HOAG G E, et al. Degradation of volatile organic compounds with thermally activated persulfate oxidation [J]. Chemosphere, 2005, 61(4): 551-560. doi: 10.1016/j.chemosphere.2005.02.032 [106] PETRI B G, WATTS R J, TSITONAKI A, et al. Fundamentals of ISCO using persulfate//In Situ Chemical Oxidation for Groundwater Remediation[M]. New York: Springer, 2011:147-191. [107] KLEIKEMPER J, SCHROTH M H, SIGLER W V, et al. Activity and diversity of sulfate-reducing bacteria in a petroleum hydrocarbon-contaminated aquifer [J]. Applied & Environmental Microbiology, 2002, 68(4): 1516-1523. [108] MCGREGOR R, VAKILI F. The in situ treatment of BTEX, MTBE, and TBA in saline groundwater [J]. Remediation Journal, 2019, 29(4): 107-116. doi: 10.1002/rem.21616 [109] 王珊, 李珊珊, 延卫. 甲基叔丁基醚生物降解研究进展[J]. 环境科学与技术, 2017, 40(5): 88-94. WANG S, LI S S, YAN W. Study progress on biodegradation of MTBE[J]. Environmental Science and Technology, 2017, 40(5): 88-94 (in Chinese).
-



 下载:
下载: