Abstract
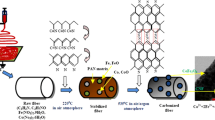
High purity Fe3O4/helical carbon nanofiber composites were obtained on a large scale by the catalytic pyrolysis of ferrocene in the presence of tin powder at 500°C over 12 h. The sizes of Fe3O4 nanoparticles are 35–65 nm in size, and the diameters of the helical carbon nanofibers range from 40–70 nm. The shapes and compositions of the nanocomposites are simply controlled by adjusting the reaction temperatures. On the basis of the obtained experimental results the formation of the helical Fe3O4/carbon nanofiber composites was investigated and discussed. The magnetic hysteresis loop of the products shows ferromagnetic behavior with saturation magnetization (M s), remanent magnetization (M r) and coercivity (H c) values of ca. 29.8 emu/g, 9.6 emu/g and 306.6 Oe, respectively.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Iijima S. Helical microtubules of graphitic carbon. Nature, 1991, 354: 56–58
Liu D Y, Luo Q M, Wang H X, et al. Direct synthesis of micro-coiled carbon fibers on graphite substrate using co-electrodeposition of nickel and sulfur as catalysts. Mater Design, 2009, 30: 649–652
Zheng T L, Wang Y H, Zheng K Y, et al. Electromagnetism and absorptivity of the modified micro-coiled chiral carbon fibers. Chinese J Aeronaut, 2007, 20: 559–563
Qin Y H, Zhang Y H, Sun X. Synthesis of helical and straight carbon nanofibers by chemical vapor deposition using alkali chloride catalysts. Microchim Acta, 2009, 164: 425–430
Yu L Y, Sui L N, Qin Y, et al. Low-temperature synthesis of carbon nanofibers by decomposition of acetylene with a catalyst derived from cupric nitrate. Chem Eng J, 2008, 144: 514–517
Kong Q H, Zhang J H. Synthesis of straight and helical carbon nanotubes from catalytic pyrolysis of polyethylene. Polym Degrad Stabil, 2007, 92: 2005–2010
Nitze F, Abou-Hamad E, Wagberg T. Carbon nanotubes and helical carbon nanofibers grown by chemical vapour deposition on C-60 fullerene supported Pd naniparticles. Carbon, 2011, 49: 1101–1107
Tang N J, Wen J F, Zhang Y, et al. Helical carbon nanotubes: Catalytic particle size-dependent growth and magnetic properties. ACS Nano, 2010, 4: 241–250
Tang N J, Zhong W, Au C T, et al. Large-scale synthesis, annealing, purification, and magnetic properties of crystalline helical carbon nanotubes with symmetrical structures. Adv Funct Mater, 2007, 17: 1542–1550
Motojima S, Chen X Q. Preparation and characterization of carbon microcoils. Bull Chem Soc Japan, 2007, 80: 449–455
Yang S M, Chen X Q, Motojima S. Coiling-chirality changes in carbon microcoils obtained by catalyzed pyrolysis of acetylene and its mechanism. Appl Phys Lett, 2002, 81: 3567–3569
Singh R K, Raghubanshi H, Pandey S K, et al. Effect of admixing different cabon structural variants on the decomposition and hydrogen sorption kinetics of magnesium hydride. Int J Hydrog Energy, 2010, 35: 4131–4137
Zhang L, Li F. Helical nanocoiled and microcoiled carbon fibers as effective catalyst supports for electrooxidation of methanol. Electrochem Acta, 2010, 55: 6695–6702
Ren X, Zhang H, Cui Z L. Acetylene decomposition to helical carbon nanofibers over supported copper catalysts. Mater Res Bull, 2007, 42: 2202–2210
Motojima S, Chen X Q. Preparation and characterization of carbon microcoils. Bull Chem Soc Japan, 2007, 80: 449–455
Luo T, Liu J W, Chen L Y, et al. Synthesis of helically coiled carbon nanotubes by reducing ethyl ether with metallic zinc. Carbon, 2005, 43: 755–759
Yang S M, Chen X Q, Motojima S, et al. Morphology and micro structure of spring-like carbon micro-coils/nano-coils prepared by catalytic pyrolysis of acetylene using Fe-containing alloy catalysts. Carbon, 2005, 43: 827–834
Bajpai V, Dai L M, Ohashi T. Large-scale synthesis of perpendicularly aligned helical carbon nanotubes. J Am Chem Soc, 2004, 126: 5070–5071
Chen X Q, Yang S M, Motojima S. Morphology and growth models of circular and flat carbon coils obtained by the catalytic pyrolysis of acetylene. Mater Lett, 2002, 57: 48–54
Bernhauer M, Braun M, Hüttinger K J. Kinetics of mesophase formation in a stirred tank reactor and properties of the products-V. Catalysis by ferrocene. Carbon, 1994, 32: 1073–1085
Wang W, Yang K Q, Gaillard J, et al. Rational synthesis of helically coiled nanowires and nanotubes through the use of tin and indium catalysts. Adv Mater, 2008, 20: 179–182
Sun Y G, Mayers B, Xia Y N. Transformation of silver nanospheres into nanobelts and triangular nanoplates through a thermal process. Nano Lett, 2003, 3: 675–679
Umar A A, Oyama M. Formation of gold nanoplates on indium tin oxide surface: Two-dimensional crystal growth from gold nanoseed particles in the presence of poly(vinylpyrrolidone). Cryst Growth Des, 2006, 6: 818–821
Zhang J H, Du J, Qian Y T, et al. Shape-controlled synthesis and their magnetic properties of hexapod-like, flake-like and chain-like carbon-encapsulated Fe3O4 core/shell composites. Mater Sci Eng B, 2010, 170: 51–57
Zhang J H, Kong Q H, Lu W L, et al. Synthesis, characterization and magnetic properties of near monodisperse Fe3O4 sub-microspheres. Chinese Sci Bull, 2009, 54: 2434–2439
Zhang J H, Du J, Ma D K, et al. One-dimensional chain Fe3O4 nanoparticles encapsulated in worm-shaped carbon shell. Solid State Commun, 2007, 144: 168–173
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is published with open access at Springerlink.com
Rights and permissions
This article is published under an open access license. Please check the 'Copyright Information' section either on this page or in the PDF for details of this license and what re-use is permitted. If your intended use exceeds what is permitted by the license or if you are unable to locate the licence and re-use information, please contact the Rights and Permissions team.
About this article
Cite this article
Zhang, J., Wang, J., Wang, H. et al. Synthesis and magnetic properties of Fe3O4/helical carbon nanofiber nanocomposites from the catalytic pyrolysis of ferrocene. Chin. Sci. Bull. 56, 3199–3203 (2011). https://doi.org/10.1007/s11434-011-4653-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11434-011-4653-2