Abstract
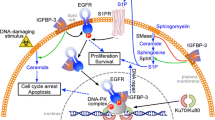
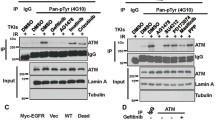
Many human tumor cells are characterized by overexpression or mutation of epidermal growth factor receptor (EGFR). Emerging evidence indicates that EGFR, as well as some of its downstream components, can translocate to the nucleus and play roles in transcriptional regulation, signaling conduction and repair of DNA double strands breaks (DSBs). EGFR in its nuclear manifestation promotes DSB repair by interacting with proteins including DNA-PK, ATM, Rad51 and BRCA1, involved in DSB repair, via the PI3K-Akt and Ras-Raf-MAPK pathways. DNA damage repair in tumor cells is emerging as an attractive target in radiotherapy and chemotherapy. Interruption of EGFR functions, or those of its downstream components, presents a promising strategy for confounding DNA damage repair in tumor cells.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Mukherjee B, Choy H, Nirodi C. Targeting nonhomologous end-joining through epidermal growth factor receptor inhibition: Rationale and strategies for radiosensitization. Semin Radiat Oncol, 2010, 20: 250–257
David J C, Chaitanya S N. The epidermal growth factor receptor: A role in repair of radiation-induced DNA damage. Clin Cancer Res, 2007, 13: 6555–6560
Dittmann K, Mayer C, Rodemann1 H P. Nuclear EGFR as novel therapeutic target. Insights into nuclear translocation and function. Strahlenther Onkol, 2010, 186: 1–6
Tao Y G, Song X, Deng X Y, et al. Nuclear accumulation of epidermal growth factor receptor and acceleration of G1/S stage by Epstein-Barr-encoded oncoprotein latent membrane protein 1. Exp Cell Res, 2005, 303: 240–251
Szumiel I. Epidermal growth factor receptor and DNA double strand break repair: The cell’s self-defence. Cell Signal, 2006, 18: 1537–1548
Dittmann K, Mayer C, Kehlbach R, et al. Radiation-induced caveolin-1 associated EGFR internalization is linked with nuclear EGFR transport and activation of DNA-PK. Mol Cancer, 2008, 7: 69–78
Dittmann K, Mayer C, Fehrenbacher B, et al. Radiation-induced epidermal growth factor receptor nuclear import is linked to activation of DNA-dependent protein kinase. J Biol Chem, 2005, 280: 31182–31189
Liccardi G, Hartley J A, Hochhauser D. EGFR nuclear translocation modulates DNA repair following cisplatin and ionizing radiation treatment. Cancer Res, 2011, 71: 1–12
Wang Y N, Yamaguchi H, Hsu J M. Nuclear trafficking of the epidermal growth factor receptor family membrane proteins. Oncogene, 2010, 29: 3997–4006
Kriegs M, Kasten-Pisula U, Rieckmann T. The epidermal growth factor receptor modulates DNA double-strand break repair by regulating non-homologous end-joining. DNA Repair, 2010, 9: 889–897
Bandyopadhyay D, Mandal M, Adam L, et al. Physical interaction between epidermal growth factor receptor and DNA-dependent protein kinase in mammalian cells. J Biol Chem, 1998, 273: 1568–1573
Hsu S C, Miller S A, Wang Y. Nuclear EGFR is required for cisplatin resistance and DNA repair. Am J Transl Res, 2009, 1: 249–258
Shintani S, Li C, Mihara M, et al. Enhancement of tumor radioresponse by combined treatment with gefitinib (Iressa, ZD1839), an epidermal growth factor receptor tyrosine kinase inhibitor, is accompanied by inhibition of DNA damage repair and cell growth in oral cancer. Int J Cancer, 2003, 107: 1030–1037
Tanaka T, Munshi A, Brooks C, et al. Gefitinib radiosensitizes non-small cell lung cancer cells by suppressing cellular DNA repair capacity. Clin Cancer Res, 2008, 14: 1266–1273
Das A K, Chen B P, Story M D, et al. Somatic mutations in the tyrosine kinase domain of epidermal growth factor receptor (EGFR) abrogate EGFR-mediated radioprotection in non-small cell lung carcinoma. Cancer Res, 2007, 67: 5267–5274
Mukherjee B, McEllin B, Camacho C V. EGFRvIII and DNA double-strand break repair: A molecular mechanism for radioresistance in glioblastoma. Cancer Res, 2009, 69: 4252–4259
Toulany M, Kehlbach R, Florczak U, et al. Targeting of AKT1 enhances radiation toxicity of human tumor cells by inhibiting DNA-PKcs-dependent DNA double-strand break repair. Mol Cancer Ther, 2008, 7: 1772–1781
Golding S E, Morgan R N, Adams B R. Pro-survival AKT and ERK signaling from EGFR and mutant EGFRvIII enhances DNA double-strand break repair in human glioma cells. Cancer Biol Ther, 2009, 8: 730–738
Bozulic L, Surucu B, Hynx D, et al. PKBa/Akt1 acts downstream of DNA-PK in the DNA double strand break response and promotes survival. Mol Cell, 2008, 30: 203–213
Chen B P, Uematsu N, Kobayashi J, et al. Ataxia telangiectasia mutated (ATM) is essential for DNA-PKcs phosphorylations at the Thr-2609 cluster upon DNA double strand break. J Biol Chem, 2007, 282: 6582–6587
Weidhaas J B, Eisenmann D M, Justin M, et al. A conserved RAS/mitogen-activated protein kinase pathway regulates DNA damage-induced cell death postirradiation in radelegans. Cancer Res, 2006, 66: 10434–10438
Toulany M, Baumann M, Rodemann H P. Stimulated PI3K-Akt signaling mediated through ligand or radiation-induced EGFR depends indirectly, but not directly, on constitutive K-ras activity. Mol Cancer Res, 2007, 5: 863–872
Toulany M, Kasten-Pisula U, Brammer I. Blockage of epidermal growth factor receptor-phosphatidylinositol 3-kinase-AKT signaling increases radiosensitivity of K-RAS mutated human tumor cells in vitro by affecting DNA repair. Clin Cancer Res, 2006, 12: 4119–4126
Meyn R E, Munshi A, Haymach J V. Receptor signaling as a regulatory mechanism of DNA repair. Radiother Oncol, 2009, 92: 316–322
Golding S E, Rosenberg E, Neill S, et al. Extracellular signal-related kinase positively regulates ataxia telangiectasia mutated, homologous recombination repair, and the DNA damage response. Cancer Res, 2007, 67: 1046–1053
Nyati M K, Feng F Y, Maheshwari D, et al. Ataxia telangiectasia mutated down-regulates phospho-extracellular signal-regulated kinase 1/2 via activation of MKP-1in response to radiation. Cancer Res, 2006, 66: 11554–11559
Li L P, Wang H, Eddy S, et al. Erlotinib attenuates homologous recombinational repair of chromosomal breaks in human breast cancer cells. Cancer Res, 2008, 68: 9141–9146
Wang S C, Nakajima Y, Yu Y L, et al. Tyrosine phosphorylation controls PCNA function through protein stability. Nat Cell Biol, 2006, 8: 1359–1368
Author information
Authors and Affiliations
Corresponding authors
Additional information
This article is published with open access at Springerlink.com
Rights and permissions
This article is published under an open access license. Please check the 'Copyright Information' section either on this page or in the PDF for details of this license and what re-use is permitted. If your intended use exceeds what is permitted by the license or if you are unable to locate the licence and re-use information, please contact the Rights and Permissions team.
About this article
Cite this article
Lu, J., Yang, L., Tao, Y. et al. Role of epidermal growth factor receptor in DNA damage repair. Chin. Sci. Bull. 56, 3132–3137 (2011). https://doi.org/10.1007/s11434-011-4650-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11434-011-4650-5